ISO 13485 and ISO 9001 for Medical Devices: How They Differ By understanding the differences between these two standards, you learn where device manufacturers need to raise the bar on quality. What many medical device manufacturers fail to realize, however, is that comparing ISO 9001 and ISO 13485 is a valuable exercise. For ISO 9001, certificates to the 2008 version expire on September 14, 2018, with ISO 13485:2003 certificates good until February 28, 2019.Ĭompanies should actively be planning their transition, learning about new requirements, assessing gaps and conducting informal audits so they can get as close to certification as possible on the first visit from external auditors.ISO 13485 is a quality system for the medical device industry, and it effectively covers ISO 9001 with some additional requirements. Meeting ISO Compliance DeadlinesĬompliance dates are coming up soon, so organizations need to be on top of the coming changes. If you’re selling devices internationally, ISO 13485 is a natural choice because it is currently being adopted as a standard by many regulatory agencies globally. Not surprisingly, some device manufacturers are opting to drop the ISO 9001 in favor of ISO 13485, maintaining a medical device-specific QMS. In general, however, medical device manufacturers will want to choose one or the other, as their structural differences would make it complicated to certify to both. For example, organizations providing related services such as contract manufacturers, suppliers or distributors might certify to ISO 13845 in addition to maintaining ISO 9001 compliance. Situations where you might consider certifying to both include when medical devices represent just part of your business. Regulatory compliance: ISO 13485 is closely linked to regulatory requirements, especially concerning complaint handling, regulatory notifications and post-market surveillance. Risk management Organizations certifying to the medical device standard will also need to show how they incorporate risk management principles into product realization and post-market feedback. ISO 13485, on the other hand, only requires demonstrating effective implementation and maintenance of the quality system.ĭocumentation: ISO 13485’s documentation requirements are much more extensive than those in ISO 9001. These differences go beyond just not sharing the same structure and include:Īims and outcomes: ISO 9001 requirements are skewed heavily towards ensuring customer satisfaction, while ISO 13485 puts more emphasis on the safety and efficacy of medical devices.Ĭontinuous improvement focus: ISO 9001 now requires manufacturers to show continuous improvement. QMS requirements: To comply with either standard, organizations will require effective processes and tools for Document Control, Employee Training, Audits and Corrective Action.ĭifferences Between ISO 9001 and ISO 13845Įven though they are similar in many ways, it’s important to note there are significant ways in which they are different.Customer focus: Both ISO 9001 and ISO 13485 are built around ensuring customer expectations are met.Plan-Do-Check-Act: Despite the fact that the two standards do not share the same structure, they both use the Plan-Do-Check-Act process approach.Risk mitigation: More so than previous versions, both standards emphasize the need for organizations to incorporate risk into design and production.ISO 9001 also shares other similarities with ISO 13485, such as: When you get down to brass tacks, ISO 9001 and ISO 13485 are both essentially about the same thing: helping companies create consistently safe, high quality products. Similarities Between ISO 9001 and ISO 13485

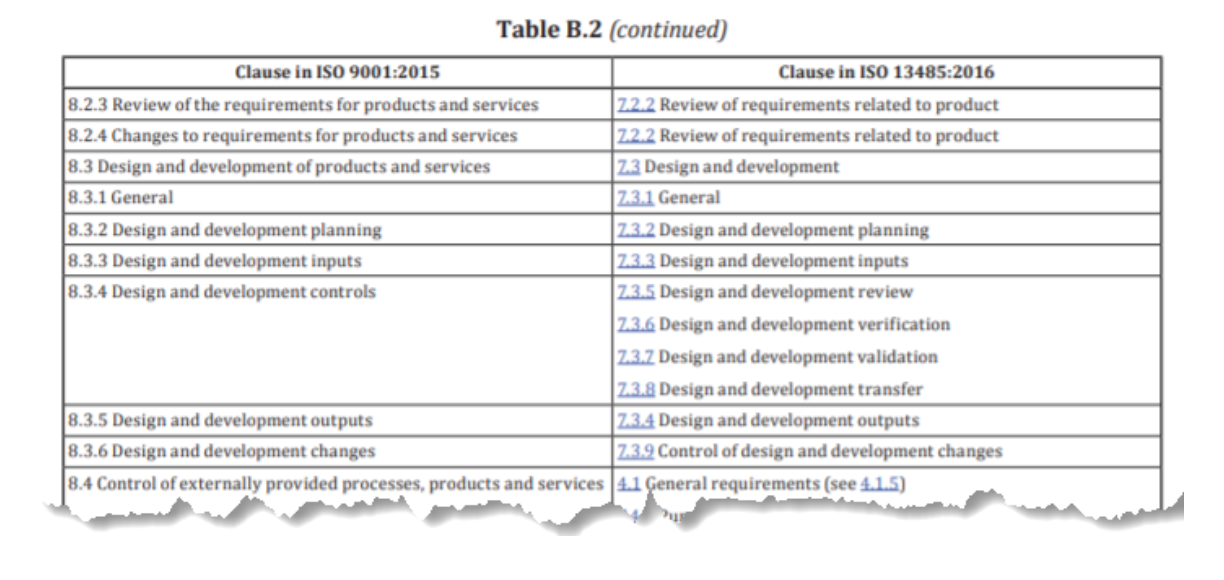
Today we’re looking at the similarities and differences between the two standards, and whether life sciences companies and related services need both certifications. Clearly there are structural differences, but you may be wondering-how different are they?

While most standard revisions now follow ISO 9001’s new high-level structure, ISO 13485 does not, even though it was released after ISO 9001. ISO 13485 for medical device quality management shares many similarities with ISO 9001, the leading global quality management standard with more than 1.1 million certificates worldwide.
